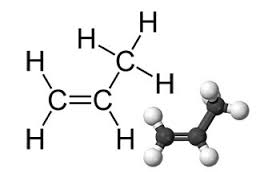
propylene
The name of the substance (Persian language): Propane
Name of matter (English): Propane
Brand (Persian): Propylene
Brand name: propylene
Other names: Methyl Ethanol, Methyl Ethylene
Packing: Carrying with special tanks
Propane is a hydrocarbon family of alkanes. Propane is a colorless, odorless gas with the chemical formula C3H6. The most important application of Propane is the production of polypropylene. Gas Propane is flammable.
Indications:
Propane is used to prepare isopropyl alcohol, polypropylene, acrylonitrile, propylene oxide, acrylic acid.
Physical and chemical properties:
Molecular formula: C3H6
Molar mass: 42.08 g / mol
Appearance colorless gas
Density: 1.81 kg / m3, gas (1.013 bar, 15C) 613.9 kg / m3, liquid
Melting point - 185.2 C (88.0 K)
Boiling point: - 47.6 C (225.5 K)
Solubility in water 0.61 g / m3
Viscosity 8.34 at 16.7C
Structure Dipole moment 0.366 D (gas)
Flashpoint -108 C
Production methods:
Propylene can be obtained from the distillation of a component of crude oil, natural gas purification or cracking of large hydrocarbons. Propylene The raw material for polypropylene production is one of the most used polymers in packaging. Most propane is polymerized using a Ziegler-Natta catalyst that produces isotactic polymers.
Chemical reactions:
Propylene-related reactions include: 1- Polymerization 2- Oxidation 3- Halogenation 4- Hydrohalogenation 5. Alkylation 6- Hydration 7. Hydrofracking
Safety Information:
Propylene gas is safe for the eyes and skin, but liquid propylene may cause freezing and burning. This gas is flammable and its mixture of air may explode.